I’ve titled this article “carbides in K390” with the idea that it will be part of a series that aims to help understand the role of carbides in knife performance and sharpening. The first article in this series, Carbides in Maxamet, dispelled the myth that carbides are weakly bonded to the surrounding steel matrix. Of particular interest in this article is the poorly understood interaction between relatively coarse (40-50 micron) grit stones and the 1-2 micron carbides found in powder metallurgy steels. As usual, we should be cautious about generalizing these observations to all examples of K390 or similar steels, or jumping to conclusions about how these microscopic observations translate to macroscopic performance. However, the observations I show here are consistent with those I have made in other samples – while they may not be the rule, they are definitely not the exception.
The particular knife used in this article is a Spyderco Endela in K390 that I purchased from Blades Canada. K390 is one of the currently popular high (9%) vanadium powder metallurgy steels. With a carbide volume fraction of around 17%, carbides in or at the apex play a major roll in how a knife made from this steel will perform and will affect the sharpening protocols required to maximize that carbide-enhanced performance.
The factory edge shows the typical morphology of carbides exposed by buffing. Grinding lines are visible in the bevel 20 microns away from the apex. Exposed carbides with matrix shadow trails dominate the last 20 microns of the bevel.



The factory edge performed adequately, slicing paper cleanly.


High vanadium steels like K390 can be challenging to sharpen as vanadium carbides are harder than typical aluminum oxide abrasive and grind/wear relatively slowly. In particular, this wear-resistance may lead to difficulty with burr formation as the apex is more likely to bend away from the stone rather than being cleanly abraded. This repeated bending of the apex may also weaken the steel, leading to micro-chipping. It is sometimes claimed that these micron-scale carbides have minimal impact on coarse grinding, with unsubstantiated claims that coarse grit stones can simply scoop out or “pop-out” the carbides. The observations I show here suggest that diamond and silicon carbide are able to abrade the vanadium carbides and remove steel sufficiently well to form a new apex. The cut depth is less than the typical carbide diameter – there is no evidence that carbides are scooped or popped out during sharpening.
Shown below is the K390 blade sharpened on a DMT Coarse diamond plate, where a moderate burr was formed. This the default result where minimal effort is made to avoid/remove the burr. This particular burr can be felt when brushing fingers off the edge. Cracked carbides are visible near the end of the burr, presumably broken during repeated flexing of the apex.


Another interesting observation is that the carbide near the surface appears to be pulverized and partially smeared out in the direction of sharpening while carbides one micron below the surface are intact.

The knife was sharpened again on the DMT Coarse, but this time with alternating, edge leading strokes, which still results in a triangular burr, although not one that can be felt or perceived in any way. Again, the part of the burr that was flexing on the diamond plate displays cracked carbides. The burnishing action of the coarse diamond plate can produce a keen edge, however the cracked carbides will likely reduce its wear resistance. This is also likely one mechanism by which microchips form.

The knife was sharpened again, this time on a Shapton Pro 320, freehand at about 33 degrees inclusive. A much smaller burr results, as compared with the diamond plate, with cleanly abraded carbides at the bevel surface. This small burr could not be detected by traditional methods.


The knife was sharpened again, this time on a Sigma Power Select II 240 grit stone at around 31 degrees inclusive angle. Once again, a relatively small burr is formed, and one that is not detectable by traditional methods. The sharper abrasives and the presence of loose particles in the mud likely minimize the burr formation as compared to the diamond plate.

Fractured carbides at the surface of the bevel suggest that the abrasion mechanism involves fracturing the carbides rather than simply abrading them or “removing them whole.”

After sharpening on a Shapton Pro 320, the knife was micro-bevelled using a translucent Arkansas stone. This type of stone generally removes steel by adhesive wear and its silicon oxide composition is much softer than the vanadium carbides in the K390 steel. Not surprisingly, it is relatively ineffective, but it does produce some interesting results. First, as with the factory edge, carbides are exposed as the matrix is wears faster than the carbides (if the carbides wear at all). Based on the amount of metal removed, this procedure has likely just exposed the carbides previously shaped by the coarse stone.


Close examination of the carbides on the micro-bevel surface show some evidence of wear (or flattening) of the carbides. There is no evidence of carbides being dislodged or “popped out.”

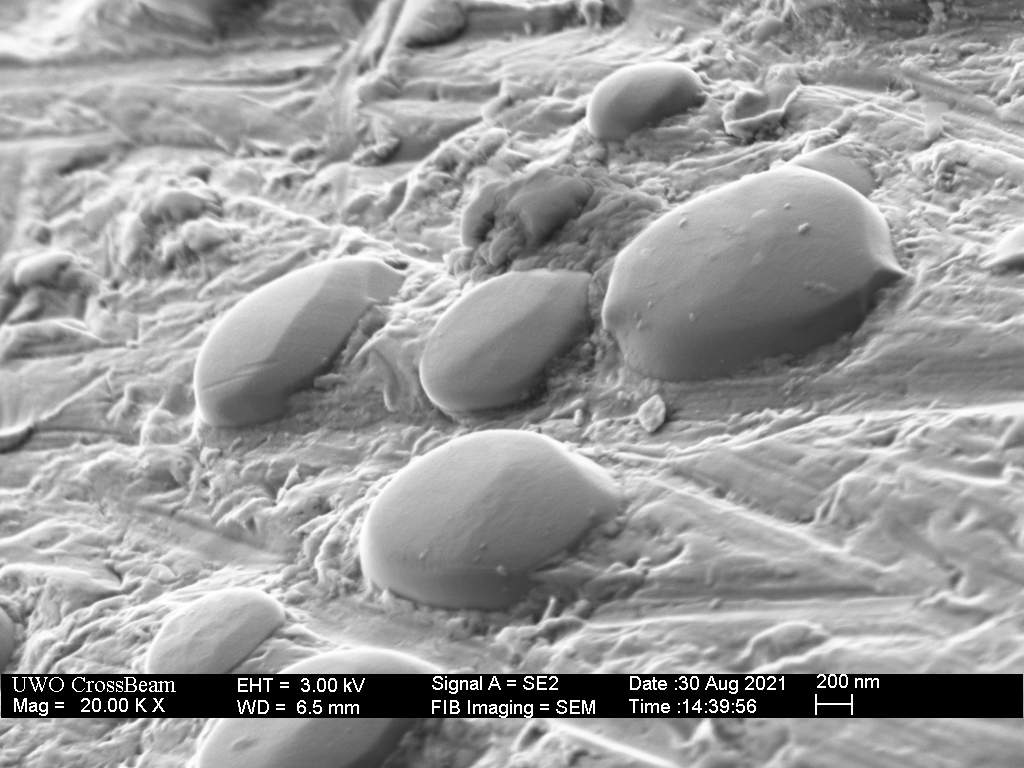
For comparison, the Spyderco Native in Maxamet steel was sharpened in the same way and micro-bevelled with the translucent Arkensas. Again, I was not able to find a hole or pit that might indicate a “popped out” carbide, but the one shown below did appear mostly excavated.

To investigate, I cross-sectioned through this particular vanadium-rich carbide and the tungsten-rich carbide on its right.


Contrary to popular belief, coarse stones do not cut deep enough to “scoop out” 1-2 micron diameter carbides in these very hard steels. Instead, the mechanism appears to be that the carbides are abraded/worn in place, flattening and thinning them until they are thin enough to shatter and be removed along with the metal swarf.

35 responses to “Carbides in K390”
Just here to say that I love this blog and tell everyone I meet interested in sharpening about it.
Thanks for the article!
LikeLike
Hi Todd,
Thank you for yet another very informative article.
How did you create the cross sections shown?
Keep up the good work, and
Cheers
John
LikeLike
Focused Ion beam
LikeLike
I own spydercos in Maximet and K 390 in Australia were I live these knives are expensive.
However the superior edge I feel is worth it.
Your writings are absolutely brilliant, ThankYou for all the dedication.
I look forward to further articles
Leith
LikeLike
Sent from my iPhone
LikeLike
Thanks again
LikeLike
You are our Myth Buster, LOL. Wow. I guess I’ll keep both my DMT and my Shapton Pro for these high Vanadium steels. It would seem there is a place for both, especially in repair / reprofiling.
Just for the sake of my curiosity, I wonder about the edge leading or trailing stroke when you sharpened on the Shapton and Sigma stones?
The surface fracturing of the carbides puzzles me, and I wonder if heat is a contributing factor?
As always, you do not disappoint!
K
LikeLike
I ended with edge leading, alternating sides to minimize the burr.
Heat is not a cause of damage, it is the energy liberated by mechanical damage. The carbides are like glass – they will break rather than bend.
LikeLike
Hi,
I recently got a S30V knife and was puzzled by how quick it dulled in what seems to be layered compressed wood (possibly chemicaly hardened too).
So I am wondering: Is this a problem of too few high hardness/toughness abrasive carbides in the steel or is the steel in general too soft and the edge just “flattens” by large scale plastic deformation effectively increasing the included angle?
In other words: would a higher “super”-carbide content help me maintain the edge longer (keeping the ~60HRC of the S30V) or would a higher HRC help me?
In other words: would K390 with a similar hardness to S30V improve the performance or would it have to be hardened to higher HRC to yield an advantage?
Thanks
Jonas
LikeLike
If you’re getting immediate dulling in resin or chemically treated wood, it’s likely rolling or chipping. Impregnated wood is much tougher on edges than just natural dried wood, and you’re left with modifying geometry at the very edge until you find something that can tolerate it.
Higher hardness might allow a finer edge, but unless your knife is really soft, adjusting geometry (just at the very apex to start) is the way to go. Carbides will improve wear resistance, but they won’t do much of anything to prevent deformation or chipping.
LikeLike
I’ve only worked with one S30V example, but you are correct that it doesn’t have a high enough percentage volume of vanadium carbides to produce a “carbide rich” edge. In my limited experience with that steel, the edge either deforms or breaks away with use, rather than wearing uniformly.
Hardness is often quoted because it’s easy to measure, but I’ve not seen any evidence that harder is better. Certainly soft steels are bad, but that doesn’t mean harder is better. There are multiple mechanisms by which an edge dulls. Deformation should be less likely in a harder steel, but micro-chipping should be more likely.
LikeLike
Hey Todd:
I just was able to give the article a
good reading – which for me is about
4 times through – and
think this is one of your best !
The photo’s are particularly nice:
they depict just what we need to see,
they are sharp with detail, and they
display the carbides so well.
The distinction between vanadium
and tungsten has never been so clear!
Once again, we are stimulated to
experiment with diamond AND
natural stones, and to think about
leading and trailing strokes……
Cheers, Scott
LikeLike
Todd: I ran your article past a friend of some 50 years that is also a motorcycle racer, and he had a couple of questions that you might be able to help with.
Here’s the first:
“Another question I have is about corrosion.
I have often seen knives become dull from
sheer exposure to the planetary atmosphere.
The sharper the edge, the more totally
surrounded by oxygen are the few atoms
that make it up. And so they eat the metal,
which yearns to return to the status of ore
in the embrace of loving oxygen.
Gradually the tip radius grows.
Those cracked carbides look like
prime locations for oxygen infiltration, too.”
LikeLike
Todd: Here’s the other question.
“The carbides in these photos are not
precipitated from the metal by heat treatment,
but are part of the initial composition of the powder, I think.
This is called “dispersed phase hardening”
and something similar has been does by
intimately mixing aluminum oxide into piston alloys
(typically by high pressure extrusion) resulting in material
with roughly double the properties
of the present day piston alloys.
F1 started down that road but the FIA
ruled that materials hardened by
a dispersed phase are composites.
So, back to reinforcing piston alloys only
with precipitated phase hardening, such as 2618.
Wonderful stuff – thank you.”
LikeLike
Thanks Todd
Awesome images and discussion.
If anyone here could point me to info dispelling the toothy edge myth I would appreciate it.
Cheeers
david
LikeLike
I’m working on a piece that will address this more directly.
LikeLike
Very excited for this. Thank you!
LikeLike
I remember Cliff Stamp once microbeveled a K390 knife with an Arkansas stone and was surprised that it worked (the knife got sharp). He hypothesized that the reason for that is that quartz is quite a bit harder than is commonly assumed. However, it seems that superior hardness is not required and the carbides can be worn and reduced by mechanisms other than cutting.
LikeLike
I don’t believe these images show evidence that the Arkansas stone cut the undamaged carbides, but I did find vanadium on the surface of the arkansas stone. I would speculate that the Arkansas stone can wear away the damaged layer (including crushed carbides) left by the coarse stone, but once it reaches “whole” carbides it stops working.
LikeLike
I have a natural stone (not Arkansas, but a similar thing – microcrystalline quartz) that works for microbeveling a 440C knife. I was always curious why that’s the case. Somehow the stone must be reducing very large chromium carbides. It does seem to me that high pressure during microbeveling is important.
If you intend this article to be a part of a series, that’s great. Chromium carbides can be very large, so investigating 440C, D2 or the like should clarify what’s going on.
LikeLike
There is a D2 instalment in the works.
LikeLike
“Fractured carbides at the surface of the bevel suggest that the abrasion mechanism involves fracturing the carbides rather than simply abrading them or “removing them whole.”
Speaking of which, sharpening crystalline materials by striking them or pressing hard into them has a very, very long history…
LikeLike
Very interesting results. Diamond fracturing hard carbides is not really a surprise, but in this very case a testament to the excellent bond of the carbide and the matrix.
As to the wearing of the carbides by the Arkansas stone that is somewhat unexpected. They do show some wear with the flattened tops and it is hard to say that it was done by the silicon oxide and not some VC on VC wear by removed carbides in the slurry.
That VC is quite the material and few things might polish it that are readily available. Titanium carbide might work.
LikeLiked by 1 person
I’m curious re: why diamond lapidary grit isn’t a good option. A few years ago, i tried a bunch of abrasives with woodworking tools and small diamonds created an edge, sharpness, and long wear in woodworking tools (unlike knives, the more refined the edge, the longer they work and the better the wear profile), and two of the steels had a lot of vanadium. they suffered nothing in terms of initial sharpness.
The conclusion (that you can better edges left by natural stones just by buying finer inexpensive synthetic diamonds) wasn’t something some people really appreciated too much. the duration results are really only applicable to woodworking, but the additional edge life from 1 micron diamonds vs. a very fine black arkansas stone was much larger than statistical noise would’ve led to.
LikeLike
These are very interesting, each new research you make on carbide seems to change how I understood many things about sharpening.
How do you think sharpening VC steel on diamond vs softer abrasive change the edge retention.
And I’m curious about the newer resin bonded diamond stone we are seeing recently, do you think they fracture the carbide less since they are supposedly less aggressive.
LikeLike
Happy to sponsor research around the resin bonded diamond stone issue. I’ve a friend who uses them in kezuroukai competitions and I’m curious!
LikeLike
I’m not sure that fracturing carbides at the surface of the bevel is a negative – it could conceivably improve the edge retention by increasing the surface hardness.
It’s something I’m still trying to understand.
LikeLike
Thank you for the great article; solid data. I’m new to sharpening and have K390 and M390 steel Spydercos. Based on the info here, would you say those can be effectively sharpened with Shapton Pro stones? Or is it still better to get diamond? As a new sharpener I’m hesitant to diamond being able to cut too fast. Appreciate the advice.
LikeLike
I’m not the author, of course, but I’ve perhaps had 500 sharpening stones and oxides down to less than a tenth of a micron.
If you’re concerned about cutting speed of diamonds, just get finer diamond from lapidary supply. It is true that diamonds will cut deeply (especially on a new electroplate hone), but it’s also true that you can mitigate the brashness by just getting finer diamonds. Graded dry diamond grit down to a tenth of a micron is pretty common and use can use it to impregnate hardwood or whatever your medium of choice with some kind of oil/solvent lubricant (like a drop of mineral oil) and follow coarse plates.
The least desirable thing about diamond hones with a knife is the feel- it’s harsh, but they work.
Look at lapidary supply for diamonds on ebay or some other similar place. 1 micron diamonds are a nice choice for finishing if you want really fine finish but still some cutting power. at the moment, I see 100 carats of diamond powder on ebay for $37 including shipping. That’s years’ worth.
You can experiment with using the fine diamonds on mild steel, well finished cast iron, hardwood, etc. The softer the substrate, the finer they’ll cut (so hardwood pulling an edge instead of pushing it to avoid carving the wood – will result in a finer edge and little or no deflection).
LikeLike
The Shapton Pro 320 is a mixture of silicon carbide and aluminum oxide and has no problem with these steels. I don’t own any higher grit Pro stones, so I can’t really comment on those. The high grit Shapton Glass do a nice job of exposing the carbides on those steels with just a bit of slurry, if you want to duplicate the factory edge. I would expect a high grit Shapton Pro to perform similarly to the Glass version.
LikeLike
I’ve got a King 1k/6k combo stone that I’ve been happily using for all my basic steels (the only kind I’d owned until recently) and haven’t really needed anything else. I can easily get VG-10 at a steep angle to glide through a tomato and my kiridashi to glide through softwood, so it was good enough. Now I’ve got K390, 3V, 10V, etc. and I’ve had the fear of the forums put into me about crushing, tearing, or otherwise ruining the fancy carbides in my blades with a softer stone. It seems like it’d be pretty silly to have my 3V camp knife with a crapped-out edge sans carbides that’ll shave hairs but burr over at the sight of canvas. I have a Spyderco cBN plate with 400/800 grit sides coming in and was looking for a complementing fine+ finishing stone or set of stones for the super steels, admitting the 800 grit cBN may be more than good enough and I’ve yet to see.
I’m a hop and skip away from JKI and the diamond 1k & 6k combo stone, which I think is a resin-matrix and not plate-type, but I wanted to see if you’d have any recommendations for someone in my position. You’d previously recommended Sigma Select II followed by DMT EF, but I’ve heard bad things about DMT (assuming one hates money).
Also, I’ve read a couple of posts and comments here about higher grit diamond plates and stones and it seems like DMT EF or Venev’s 1200 OCB is about as high as I’d want to go… until JKI stepped in. Any chance you’ve heard about or have experience with those stones, specifically the 6k one? Recommendation?
No matter the primary stones, would you follow up with a softer, finer hone, like an UF translucent or black novaculite in the spirit of ‘celebrating the carbides’ and removing some surrounding matrix? I don’t need the Sigma coarse grits given I have the cBN on its way in, but maybe the Select II 13000 for that application? If you would take this step, would you go straight here from the 800 grit cBN or is that step/are those steps in between a good idea?
FWIW I am more confused about what I should be doing after reading almost every post here and browsing the forums than when I started with only a Smith’s pocket sharpener years ago and had the task of “pull knife, pull more make sharper”. Thank you in advance for the help, and I really appreciate your site and the effort you put into your work! Although I may be confused, I’m vastly more informed.
LikeLike
Do your primary work with the diamond combo you mentioned and follow it with loose 1 or 0.5 micron diamond powder that’s about $10 on Ebay. Use it with hardwood or softer with a light oil lubricant and compare the results to your other stones.
LikeLike
I have no experience with JKI. I did analyze the Venev 1200 OCB and confirmed that the binder is not abrasive and that the diamonds are of the expected size – I don’t have any comment on it’s sharpening use at the moment though.
I’m really still trying to understand the value of these high vanadium steels and determine the best way to sharpen them.
LikeLike
I recently acquired a serrated k390 blade and am having a difficult time finding fine/Xtra fine diamond cylindrical sharpening rods to maintain the cutting edge. I found some red ruby 6000grit rods(I think are crystalline aluminum oxide with some chromium for color) but from my reading it doesn’t appear they are hard enough to hone the edge. But this research has me questioning if they might be alright to micro-bevel with and expose more intact carbides for edge retention. ? Any suggestions for sharpening serrated k390? Thanks
LikeLike
purchase diamond lapidary grit and apply it to a dowel rod as a paste with oil and wax and use the dowelrod like you would with a drawing motion rather than edge in on a steel.
K390 is 9% vanadium. It’ll sharpen (slowly!) with alumina in ruby rods, but it won’t do it the right way and perhaps may waste your time.
that said, you could still try it – much depends on what alumina will do on something like K-390 in terms of the actual hardness. If you knife is 64/65 hardness, a ruby rod is really going to struggle and the edge will be on a micro level uneven. Will it matter compared to geometry in a serrated knife? Probably not.
what I mentioned will also work with mild steel and cast iron rod – the diamond will embed into the rod, but it’s probably not that easy to find a suitable rod with a decent level of polish. cast iron works better for that than mild steel – the diamonds get stuck in the softer of the two surfaces like you’d stick something in glue, and then the harder surface slides over them.
I don’t know what finish level you’re looking for but 5 micron diamonds are a little bit harsh but fast cutting, and 1 micron and below super fine. The lapidary supply on ebay is all good stuff, or even ali express. Lapidary diamond powder.
LikeLike